Working with HF
Hydrofluoric acid (HF) has a number of chemical, physical, and toxicological properties, which make handling of this material especially hazardous. Although chemists consider HF to be a “weak” acid, its potential to produce serious health effects greatly exceeds that of “strong” acids commonly used in the laboratory. HF shares the corrosive properties common among mineral acids but is unique in its ability to cause deep tissue damage and hypocalcemia (low calcium levels in the blood). Anhydrous (containing no water) HF is a clear, colorless, fuming, and corrosive liquid. HF is also available in the gaseous state. All forms, including solutions or vapor, can cause severe burns to tissue which are extremely painful and very slow to heal.
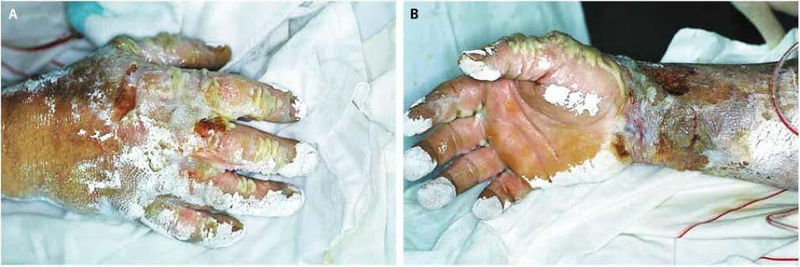
The following picture shown an example of an HF burn caused by exposure to 70% hydrofluoric acid.
Chemical properties
Hydrofluoric acid solutions are clear and colorless with a density similar to that of water. The most widely known property of HF is its ability to dissolve glass. It will also attack glazes, enamels, pottery, concrete, rubber, leather, many metals (especially cast iron) and organic compounds. Upon reaction with metals, explosive hydrogen gas may be formed. Therefore, you must use and store HF in polyethylene, polypropylene, or Teflon.
Toxicological properties
The unique toxicological properties of HF are due to the action of the fluoride ion. Fluoride ions cause soft tissue and bone damage by binding to calcium. Fluoride ions are both acutely and chronically toxic. Acute effects of HF exposure include extreme respiratory irritation, immediate and severe eye damage and pulmonary edema.
Skin contact with HF is probably the most common route of exposure for laboratory personnel, although HF can also cause damage through eye contact, inhalation, or ingestion. Exposures to concentrated (>50%) HF solutions will cause immediate, severe, penetrating burns. Exposure to less concentrated solutions may have equally serious effects, but the appearance of symptoms can be delayed for up to 8 hours for concentrations of 20-50% and up to 24 hours for concentrations less than 20% HF.
Concentrated HF burns can be fatal even if only 2% (~20 cm2) of the body is exposed. Working with anhydrous HF is extremely dangerous due to the vapors it produces, which present a severe inhalation hazard.
If you are exposed to hydrofluoric acid, seek medical attention immediately, even if you do not feel pain.
Exposure control, PPE and work practices
HF must only be used in the HF designated fume hood. NEVER use HF outside the fume hood! In order to warn and protect others from the hazard of HF, a warning sign indicating the use of HF must be posted on the fume hood. The designated HF fume hood safety zone marking on the floor ensures a safe comfort operating zone for the user. Other cleanroom users must stay outside this zone when HF is used.
Always wear two pairs of gloves (double gloves), a lab coat, rubber apron, face shield, disposable arm/sleeve covers and chemical safety goggles when working with any HF solution. Always check gloves for leaks prior to use. In case of spills on the gloves they must be replaced and dumped in the sink and flushed with copious amounts of water before being disposed as chemical waste in a labelled plastic container.
The purpose for personal protective equipment (PPE) is to shield the individual in the event of a release of vapor, a spill or other incident. PPE is not a substitute for safe work practices.
The threshold limit value (TLV) for HF is 2 PPM.
Ensure that calcium gluconate gel is available before using HF!
The use of HF requires a buddy to be present!
HF exposures
Although HF exposures can result in injury, quick response will minimize the damage. All exposures should be treated immediately even though burns may not be felt for hours. HF first aid (calcium gluconate gel) and spill response kits are located in the clean room and users are obliged to familiarize themselves with the location and proper use of them. Affected personnel must receive medical attention for all exposures. Maintain a copy of these procedures and the MSDS to take to the emergency room.
- Skin Contact – Immediately wash all affected areas with water. Be sure to remove any clothing or jewelry that could trap or retain HF (remove goggles last). Flush skin for fifteen minutes or until medical attention is available. Get medical attention. Apply calcium gluconate gel to the affected area (use rubber gloves) every fifteen minutes and repeat flush in between.
- Eye Contact – Immediately flush eyes for at least fifteen minutes with water while holding eyelids open. Get medical attention. Flushing can be limited to five minutes if medical personnel are immediately available to administer sterile calcium gluconate (1%) solution (via continuous drip into eyes).
- Inhalation – Move to fresh air as soon as possible. Get medical attention. Medical personnel can administer pure oxygen and calcium gluconate (via nebulizer) to patient. Laboratory personnel should only attempt to clean up small HF spills (pool less than 10x10 cm) that do not involve personnel contamination and that are contained and under control. Be sure that good ventilation is available and that personal protective equipment is worn before attempting to clean up a HF spill.
Using hydrofluoric acid safely
- Never use Hydrofluoric Acid when working alone or after hours. Hydrofluoric Acid may be used when working alone during normal working hours provided knowledgeable laboratory personnel have been alerted and at least one is in the general vicinity (buddy system).
- All lab personnel, not just those who will be using Hydrofluoric Acid, should be informed of the dangers of this chemical and the emergency procedures necessary in case of an accident. A sign must be posted to alert people that work with Hydrofluoric Acid is in progress.
- All persons who will be using Hydrofluoric Acid must be made aware of its properties and trained in proper procedures for use and disposal.
- Make sure that a paper copy of these guidelines and the MSDS is at hand before starting to work.
- Before beginning any procedure involving Hydrofluoric Acid, make sure the access to the emergency shower (just outside the clean room) and eyewash is unobstructed
- Only experienced persons familiar with its properties should handle the concentrated acid, and mix solutions
- A small supply of sodium bicarbonate or calcium hydroxide for spills must be kept near the hood where the work will be conducted. If a small quantity (100 ml or less) of dilute Hydrofluoric Acid solution is spilled, dike the spill with sodium bicarbonate or calcium hydroxide and clean it up by applying a thin coat of powder. Alert the clean-room staff to dispose of the residue. If a larger amount is spilled, or the acid is concentrated, contain the spill as best you can, evacuate the area, alert the clean-room staff and if needed call emergency services 112. Avoid exposure to the vapors. Do not use silica-containing agents, especially diatomaceous earth (kitty litter) vermiculite or sand, to absorb hydrofluoric acid, because they can react with HF to form toxic silicon tetrafluoride gas
- HF waste must be disposed of in the appropriate HF waste container located in the cabinet below the fume hood.
- Always use the appropriate PPE when working with >1% HF solutions
- Any exposure to Hydrofluoric Acid must be medically evaluated spill.